Overview
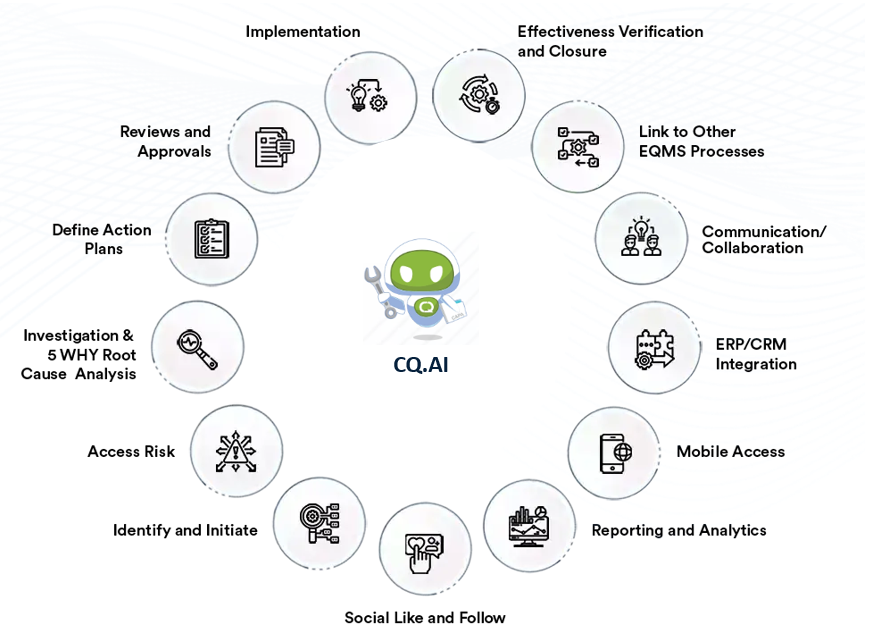
CQ CAPA tracks actions that correct issues and prevent them from recurring. You document the problem, evaluate the cause, and define actions that address both the immediate issue and underlying conditions. CAPA records create a traceable history that supports quality and compliance activities.
Use Standard CAPAs for straightforward issues that require defined corrective and preventive actions. Use 8D CAPAs when you need a structured, multi-step method that investigates causes, validates solutions, and confirms long-term effectiveness.
Additional Resources
ComplianceQuest
We Value Your Feedback
To provide feedback or suggestions to improve the help content on this page click here.
