Overview
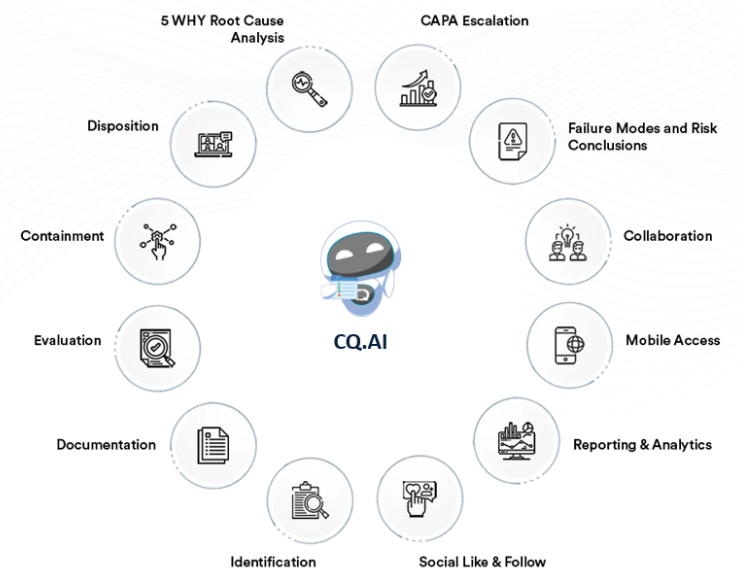
CQ Nonconformance Management records nonconforming products or processes identified during manufacturing, testing, or inspection. Users document what went wrong, where it occurred, and how the nonconformance was contained so impact can be assessed and follow‑up actions defined.
The nonconformance record can link to related complaints, investigations, CAPAs, or risk records so users can track how the issue was evaluated and resolved across quality processes.
Additional Resources
ComplianceQuest
We Value Your Feedback
To provide feedback or suggestions to improve the help content on this page click here.
