Overview
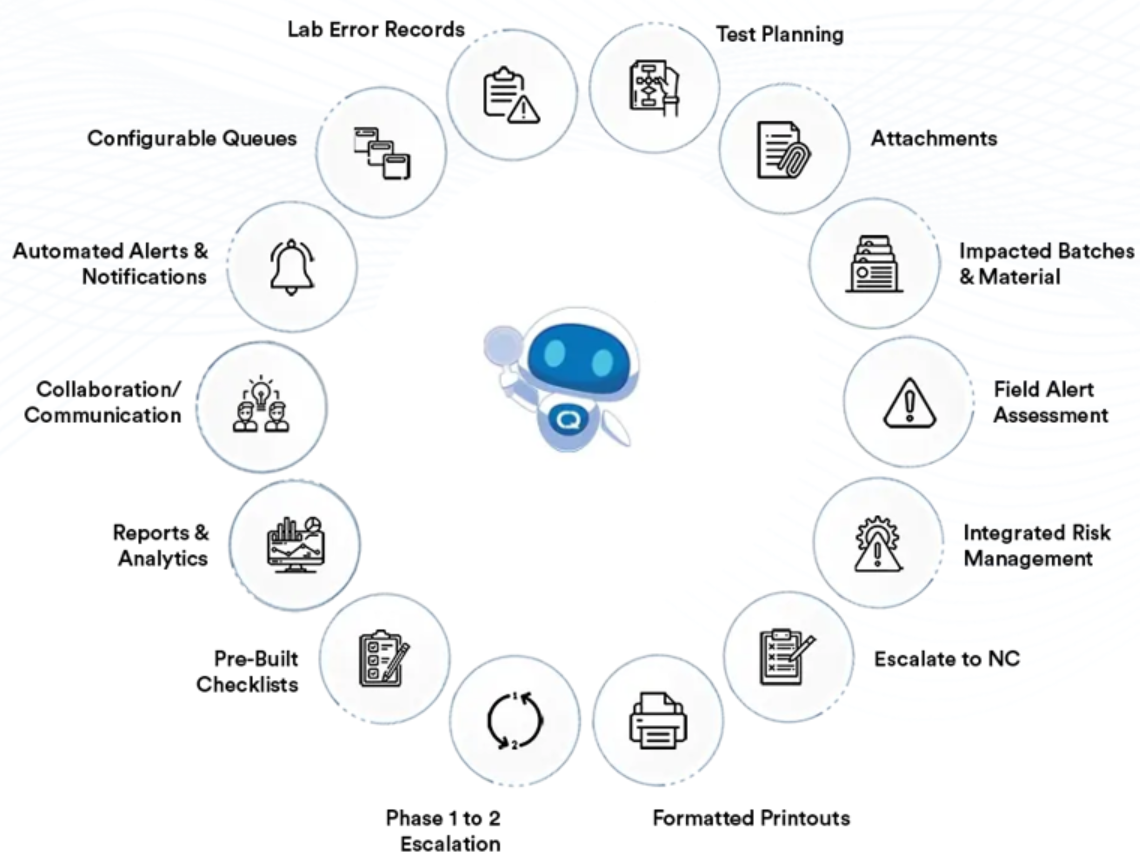
CQs Laboratory Issues (Lab OOS) ensures your investigations are systematic, comprehensive, and efficient. Collaboration tools and guided workflows make it easier for your QC and QA teams to get to the assignable or root cause of every OOS test result and then act on it, while staying compliant, thanks to thorough documentation.
CQ’s Laboratory Investigation solution allows your laboratory teams to initiate to a Phase 2 Investigation whenever necessary, conduct a full production review, then escalate to a CAPA if needed to ensure high quality products.
How to Articles
In this section, you’ll learn how to:
Each topic includes step-by-step instructions, screenshots, and best practices to help you navigate laboratory issues management efficiently and in alignment with your organization’s quality management goals.
Let’s get started with mastering the laboratory issues management process in ComplianceQuest.
Summer 2026
